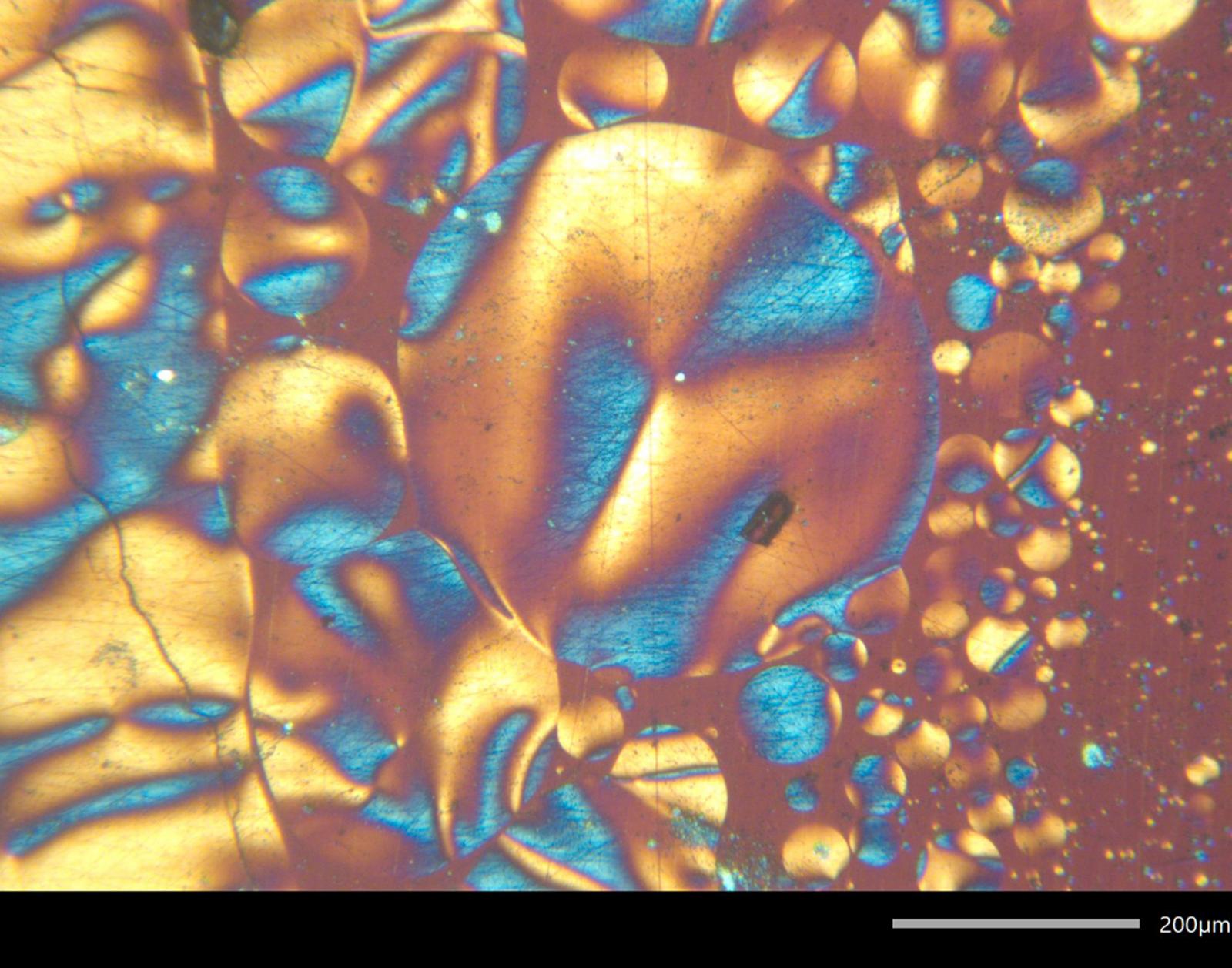
Entry #03
"Made from Scratch: Scratch Induced Birefringence in Conjugated Polymer Indacenodithiophene-co-benzothiadiazole (IDT-BT)"
Artist's Statement: This unedited image calls to mind the beautiful blue Morpho menelaus butterfly, which has strikingly blue wings that are delicate but powerful. Like the Morpho menelaus butterfly, this conjugated polymer thin film gets its color from its unique nanoscale structural properties and not because it is pigmented.

Entry #04
"Graphene x Andy Warhol"
Artist's Statement: I remember seeing the MVC winners from past years hanging in Steidle during my campus tour and thinking, "Wow, I hope one day I will be able to do something like that." Now here I am, 2 years later, submitting my own image for MVC 13. Andy Warhol is well known for his pop art, and since I grew up in Pittsburgh like him, I couldn't resist drawing the comparison to my image. My piece is authentic, straight out of the Nano Scope software. I know it will be hard to compete with the extraordinary computational and SEM images, but I firmly believe I have made the best possible submission I could with the tools and knowledge I have.
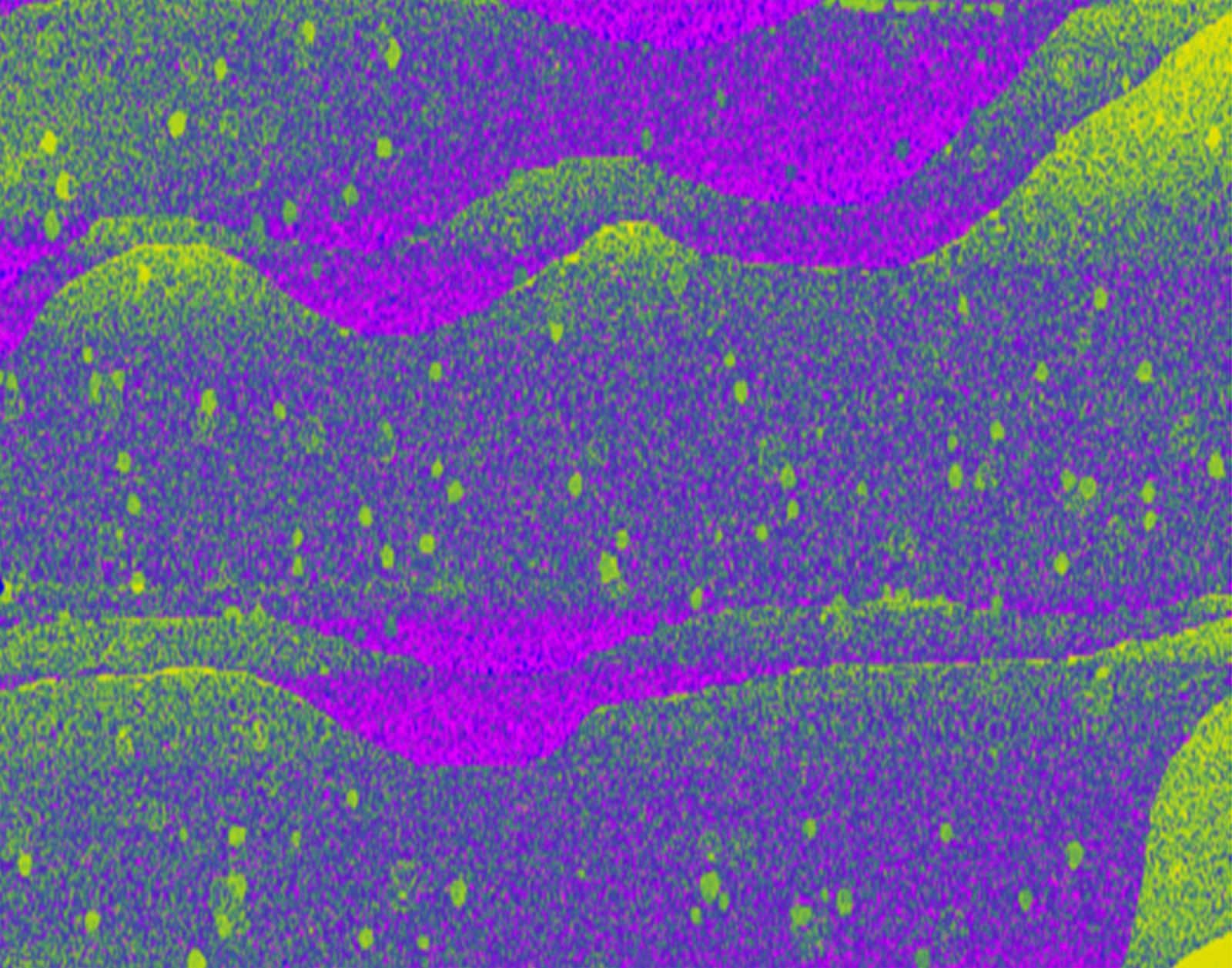
Entry #05
"Formation of a discotic liquid crystal: Nucleation, growth, and coalescence of mesophase spheres driven by the principles of chemistry and physics into a visual narrative of science with line, shape, color, texture, and space as shown in a polarized-light micrograph"
Artist's Statement: This is a microscope image of a semi-coke obtained from carbonization of a petroleum feedstock in a laboratory reactor. To the naked eye, it appears black, similar to coal. “The color black relates to the hidden, the secretive and the unknown, and as a result it creates an air of mystery.” A polarized-light microscope may uncover this mystery and liberate the colors blue, yellow, and magenta by the interaction of polarized-light with anisotropic carbons. Capturing all three stages of mesophase development in just one image offers a powerful visual narrative of the dynamic process with changes in shape, color, and texture of objects from the birth of mesophase spheres (nucleation) on the right of the image to their growth and interaction with others (coalescence), and into their full maturation into anisotropic domains on the far left. In addition, the memory of these interactions is retained with patches of yellows and blues remaining from individual spheres that were coalesced.
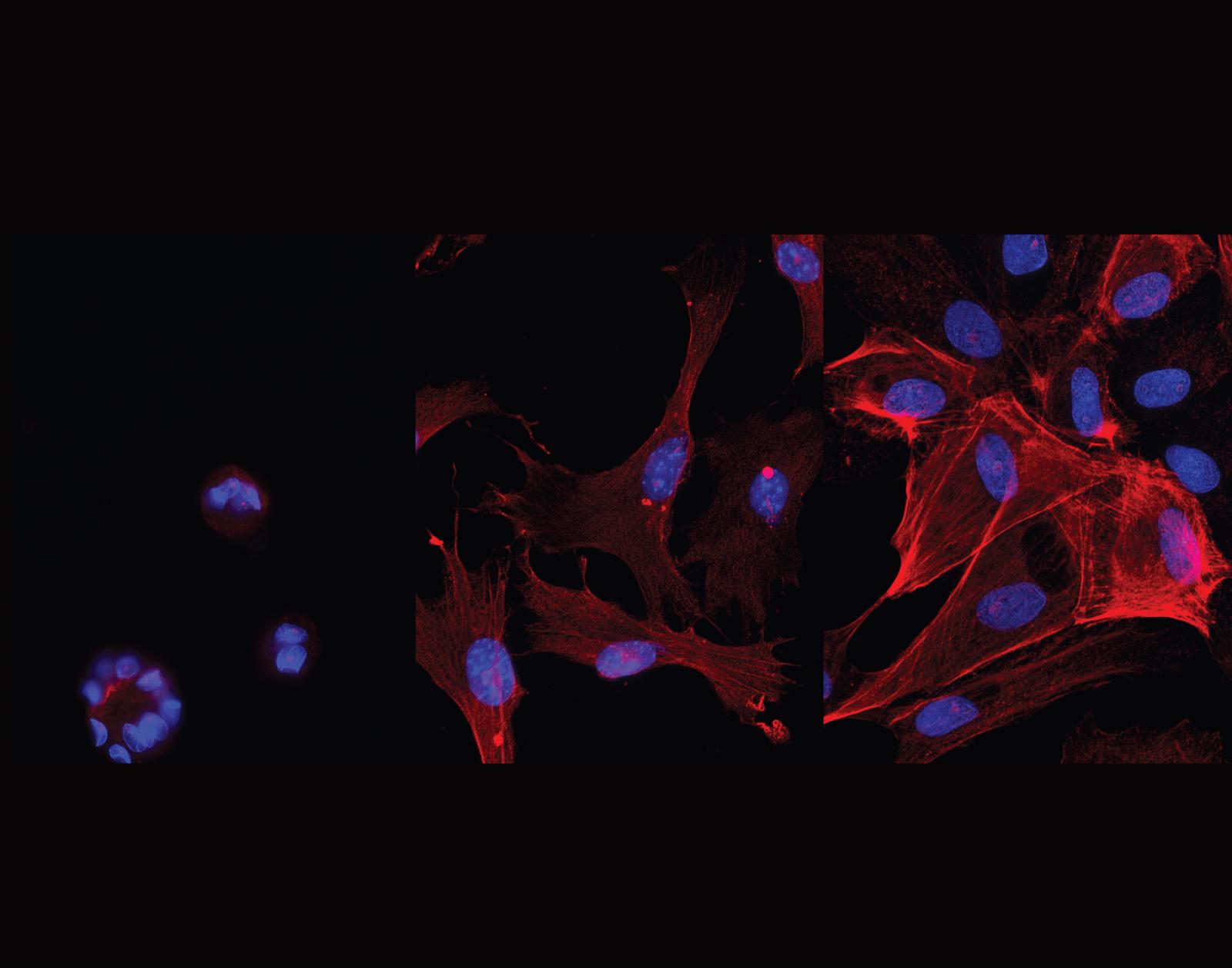
Entry #06
"Rigidity of the extracellular matrix surrounding the cell promotes Epithelial-Mesenchymal Transition"
Artist's Statement: My research is based on the big-picture of understanding how the mechanical properties of a tissue regulate cell signaling pathways. If we can better understand how cells behave in response to tissue mechanics, we can develop strategies to target the signaling pathways and in turn, negate pathological conditions such as cancer and fibrosis. My current work involves studying the effect of modulus of the cell’s extracellular matrix on chromatin architecture and how it affects gene transcription machinery during the process of Epithelial-Mesenchymal Transition (EMT). The picture I am trying to convey with the help of my image submission is that mechanical stimuli can independently regulate the expression of proteins in epithelial cells during the process of EMT. Hence, I combined fluorescence images taken for EMT-induced epithelial cells that were seeded on increasing matrix rigidities to show the audience how the increase in modulus enhances EMT-induced protein expression in cells
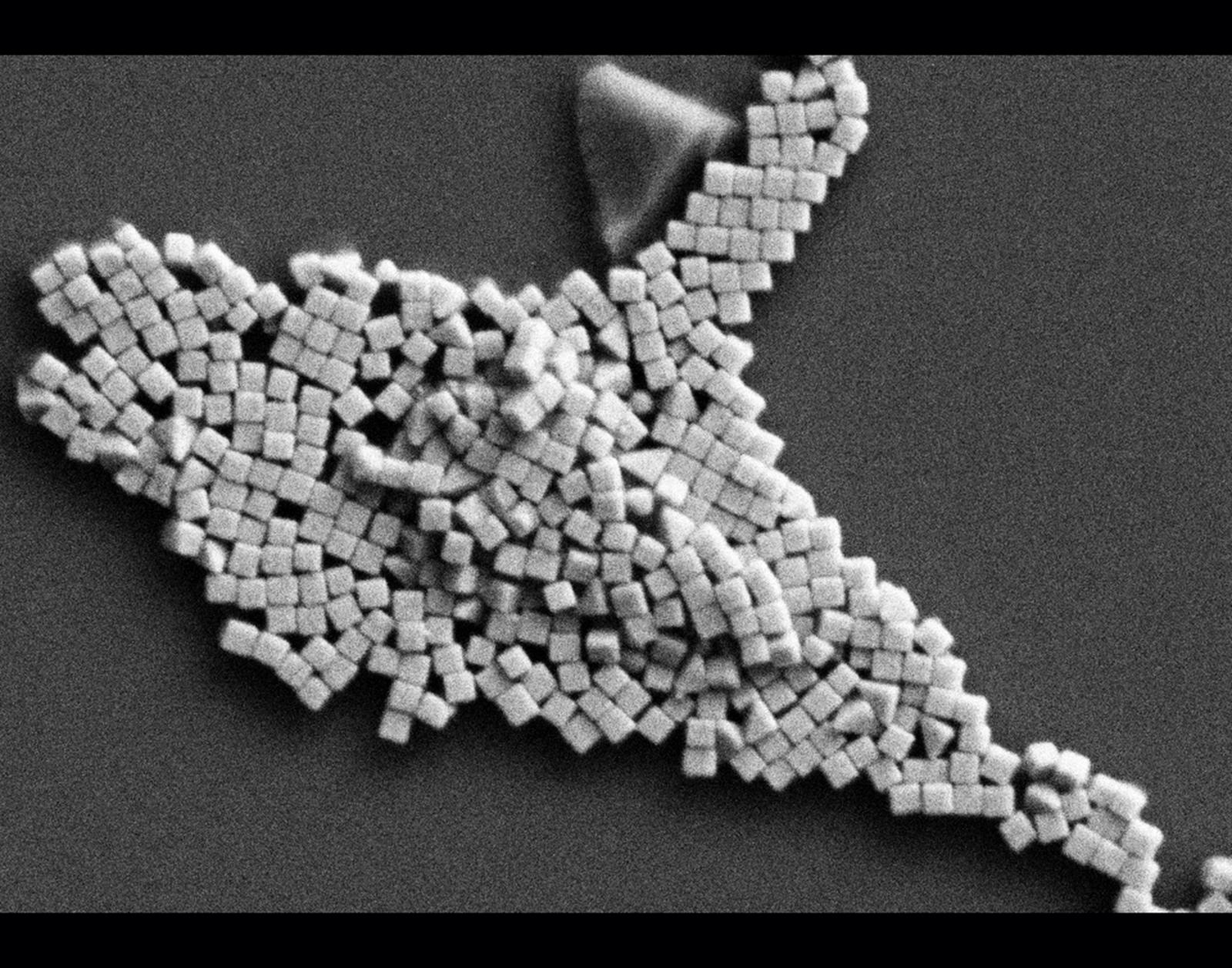
Entry #08
"Aerial View of Potassium Niobium Silicate Oxide Ice Mountains"
Artist's Statement: While the cold winter months may weigh heavy on some, I look forward to the brisk winds and billowing snow drifts that accompany winter. With my boots, hat, and coat, I welcome the invigorating cold air as I trudge up mountains and scurry across wind-swept fields. Looking at this image, I was reminded of mountain tops covered in snow and ice. While my personal views of mountains are typically limited to magnificent vistas seen from neighboring peaks, this image gave me a unique perspective as I imagined myself looking down the electron gun to enjoy an aerial view of mountains covered in ice crystals waiting to be conquered. From this perspective, snowy cliffs and exotic microstructure are not daunting, but inspiring as they present unique opportunities to explore uncharted territory.

Entry #09
"Heavenly Patterns: Widmanstätten Microstructure in Additively Manufactured Duplex Stainless Steel"
Artist's Statement: I still remember the day I first used a microscope to look at onion cells back in middle school. I was astonished at what lies beyond the view of the naked eye. Fortunately, till this day I am still in awe of the world found under a microscope. Microscopy helps us understand much more about material systems and how new sustainable means of manufacturing influence them. Additive manufacturing is a gateway to new discoveries that will eventually improve upon traditional manufacturing methods resulting in more cost-effective and advanced materials with better properties. As the perseverance rover recently landed on the surface of Mars, one day the next mission will be carrying a 3D printer capable of printing materials on the spot aiding the journey of interplanetary travel.

Entry #11
"A dual-phase microstructure of a 3D-printed stainless steel alloy"
Artist's Statement: The image shows the dual-phase microstructure of a duplex stainless steel alloy fabricated using additive manufacturing, also known as 3D printing. This dual austenite and ferrite microstructure, which etched blue and yellow respectively in the image, contributes to the high strength of these alloys. Additive manufacturing provides a processing route to produce a fine austenite grain morphology, compared to traditional wrought duplex stainless steel alloys. This fine microstructure can more effectively inhibit dislocation motion during plastic deformation, resulting in improved strength over traditional wrought products.
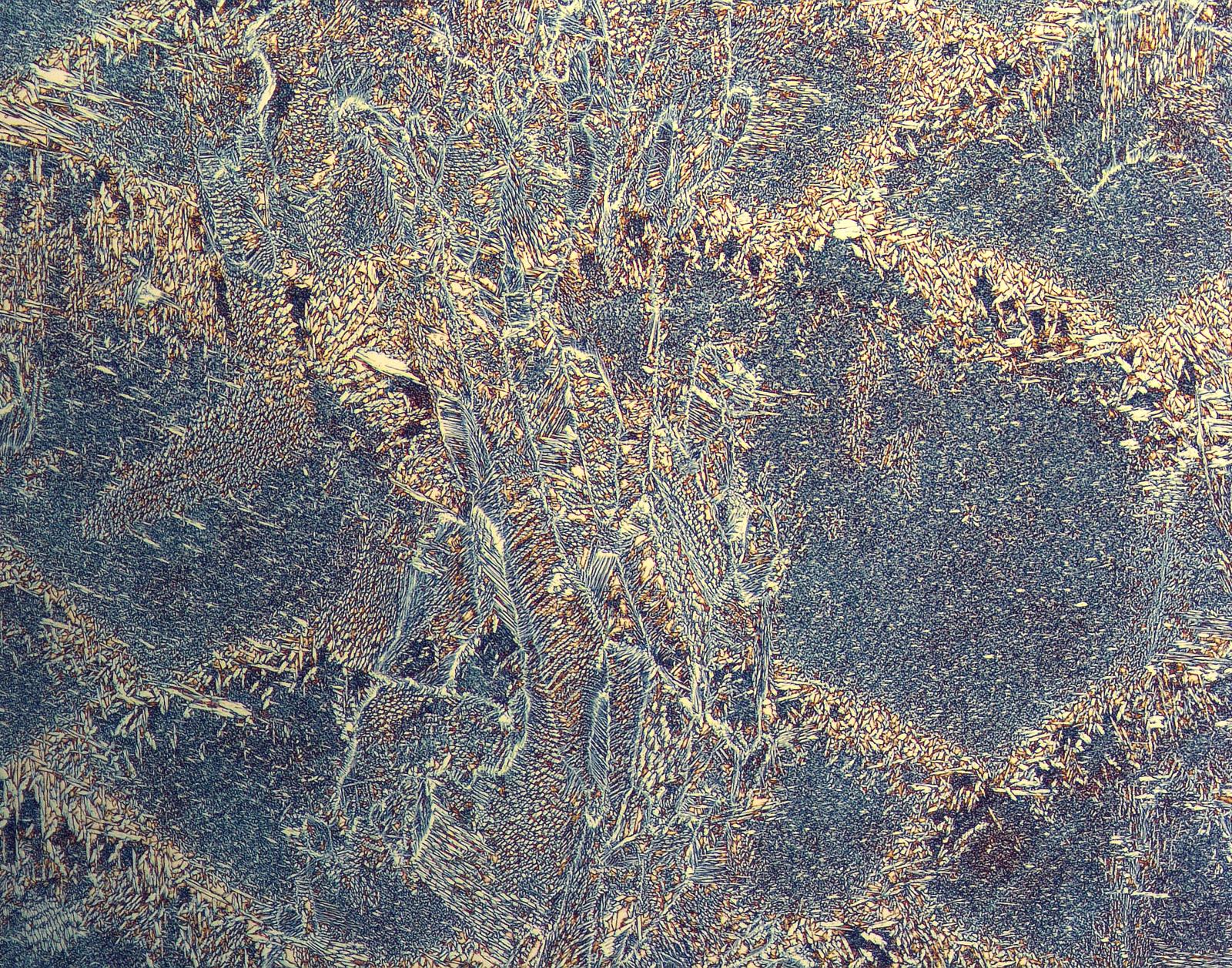
Entry #12
"Never-ending Friendship: Titanium(IV) isopropoxide (TTIP) decomposition on SrO terminated SrTiO3"
Artist's Statement: As a child of immigrants who moved from the former Yugoslavia to Turkey and now an international student in the US, the concept of friendship has been very meaningful for me my entire life. Wherever I have been, the friends who received me with open arms broadened my horizons and motivated me to discover more outside my little family. Likewise, there is a never-ending friendship between TTIP and SrO-terminated surface where TTIP discovers itself as well as its environment. That way, this tiny TTIP molecule is finally able to be a harmonious part of the atomic community in which it is more than welcome.
Entry #16
"The Shell - Additive manufactured Ti-6Al-4V fracture surface"
Artist's Statement: The spherical gaseous pores have fine shell structure with elegant whirls that initiated from the melt pool solidification on boundary of gas and solid alloys. There are both single placed shells and overlapped ones that separated all over the fracture surfaces, and their unique neat fashion make them stand out compared to the messy crack networks and failure cliffs.

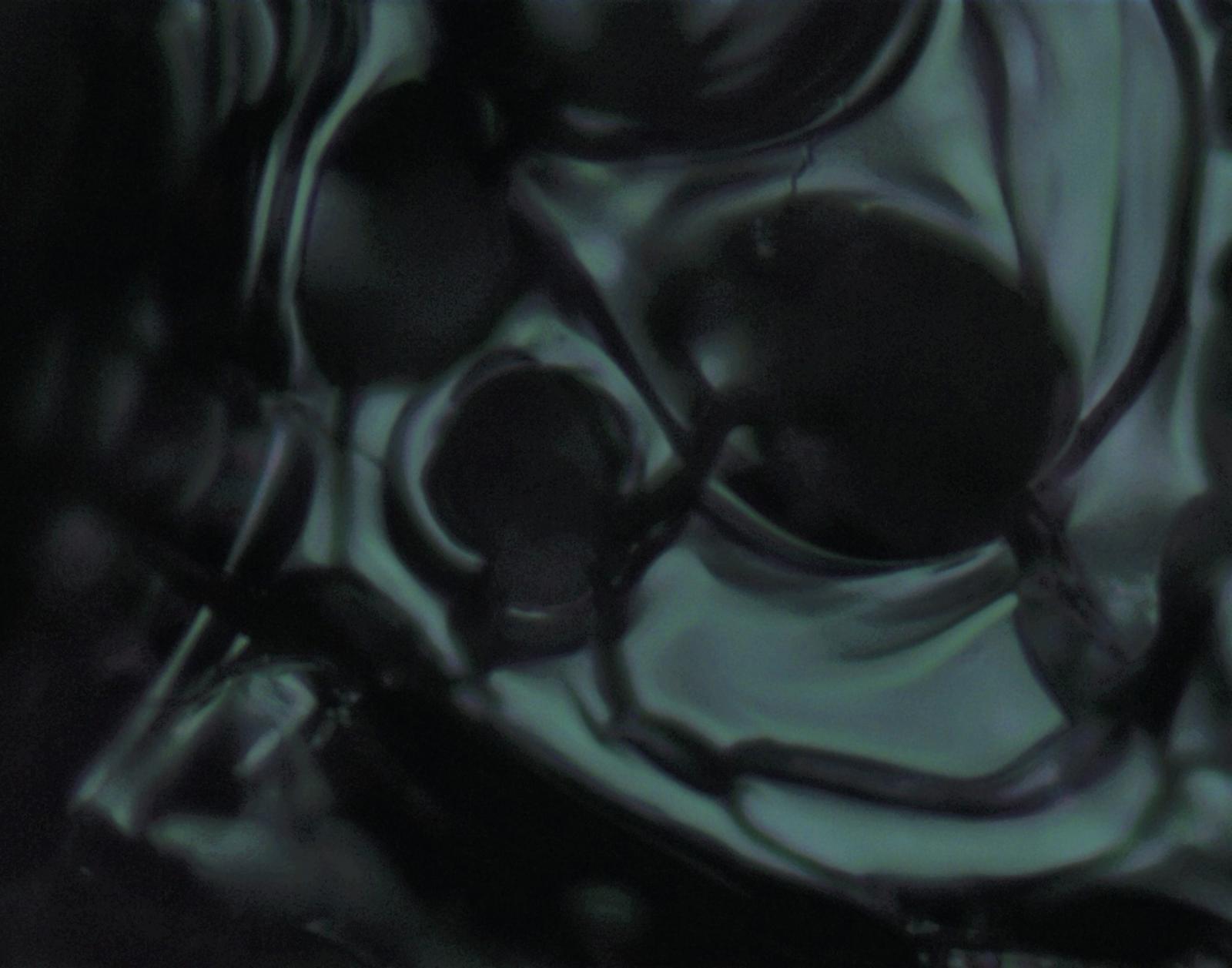
Entry #17
"Mixed Chromium Oxide in the Shape of a Skull"
Artist's Statement: As many discoveries are sometimes made, I came upon this surface defect entirely by chance during the characterization of this sample with Raman spectroscopy. Imagine my surprise while collecting a spectrum to see a skull looking back at me! The morphology of our samples created through laser-induced thermal synthesis have been well studied through other methods such as scanning electron microscopy and profilometry. This image reminds us to always keep our eyes peeled for discovery in our research, whether it’s noticing a unique surface feature while collecting a spectrum or finding new properties in a material.
Entry #18
"Chirality-induced conformational disorder in relaxor ferroelectric polymers"
Artist's Statement: We proposed a molecular understanding of disordered structure in polymeric relaxor ferroelectric materials which finds many promising applications flexible electronics, actuators and transducers, energy storage, piezoelectric sensors and electrocaloric cooling. Using atomic force microscope infrared spectroscopy, we can directly map the spatial distribution of chemical structure at the molecular level. Seeing is believing. The molecular chain conformation is found to be highly disordered in relaxor ferroelectric polymers, which strongly supports our proposed conformational disorder picture.

Entry #19
"Multimetal Particles Sintered to Glass Substrate"
Artist's Statement: When I first saw this film, I was vaguely reminded of an abstract night sky with the gray substrate resembling the vast space that was then dotted with stars (Fe/Co/Ni oxide particles in reality). As I continue my research in the Zarzar group at Penn State, I am thankful of these moments where I can repeatedly experience the awe and wonder of scientific observation.

Entry #20
"Dendrites in a Battery"
Artist's Statement: Light like a feather and delicate like a fern? Do not let the looks fool you. These metal dendrites grown from battery cycling will penetrate the separator, short-circuit the battery, and cause the battery to overheat and, in some instances, catch on fire. This image inspires me because of the soft, delicate appearance in contrast to the material's stiff nature, a candy to eyes but a poison to batteries.

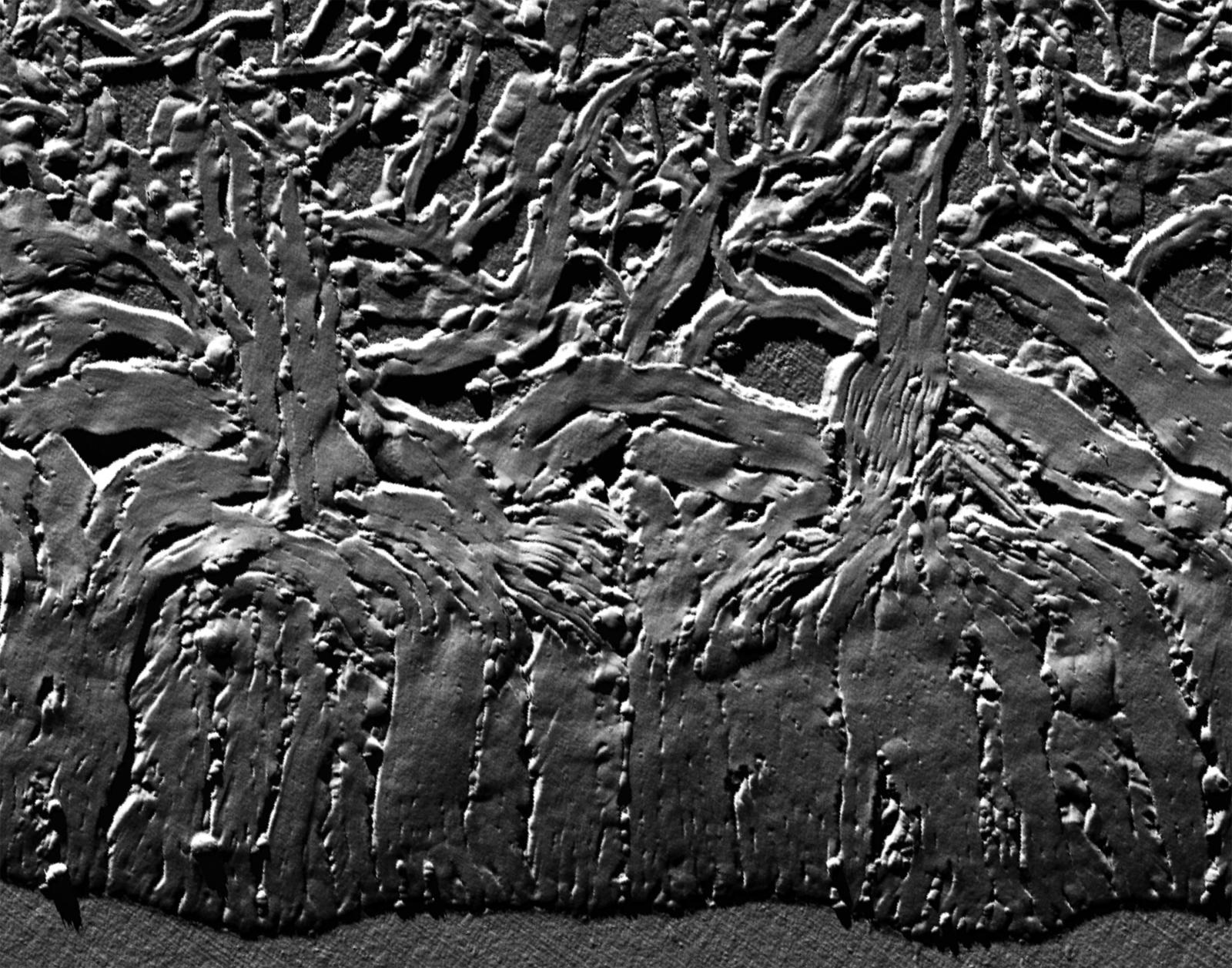
Entry #22
"Strength in Numbers: Cellulose Bundling in Plant Secondary Cell Walls Reinforces Individual Cells"
Artist's Statement: I chose this novel snapshot of cell wall formation because it depicts the bundling of cellulose in the plant secondary cell wall in a very palpable way. The formation of secondary cell walls is what allows plants to grow vertically. In this atomic force microscopy scan, the secondary cell wall bundles are evocative of tree trunks that grow upwards and then branch outwards. This web of cellulose bundles provides strength on the microscale in a way that mirrors the structure that they ultimately support on the macroscale.
Entry #23
"Different facets of unearthed carbides"
Artist's Statement: Carbides are hiding away from the melting pit and plotting a scheme to escape.

Entry #26
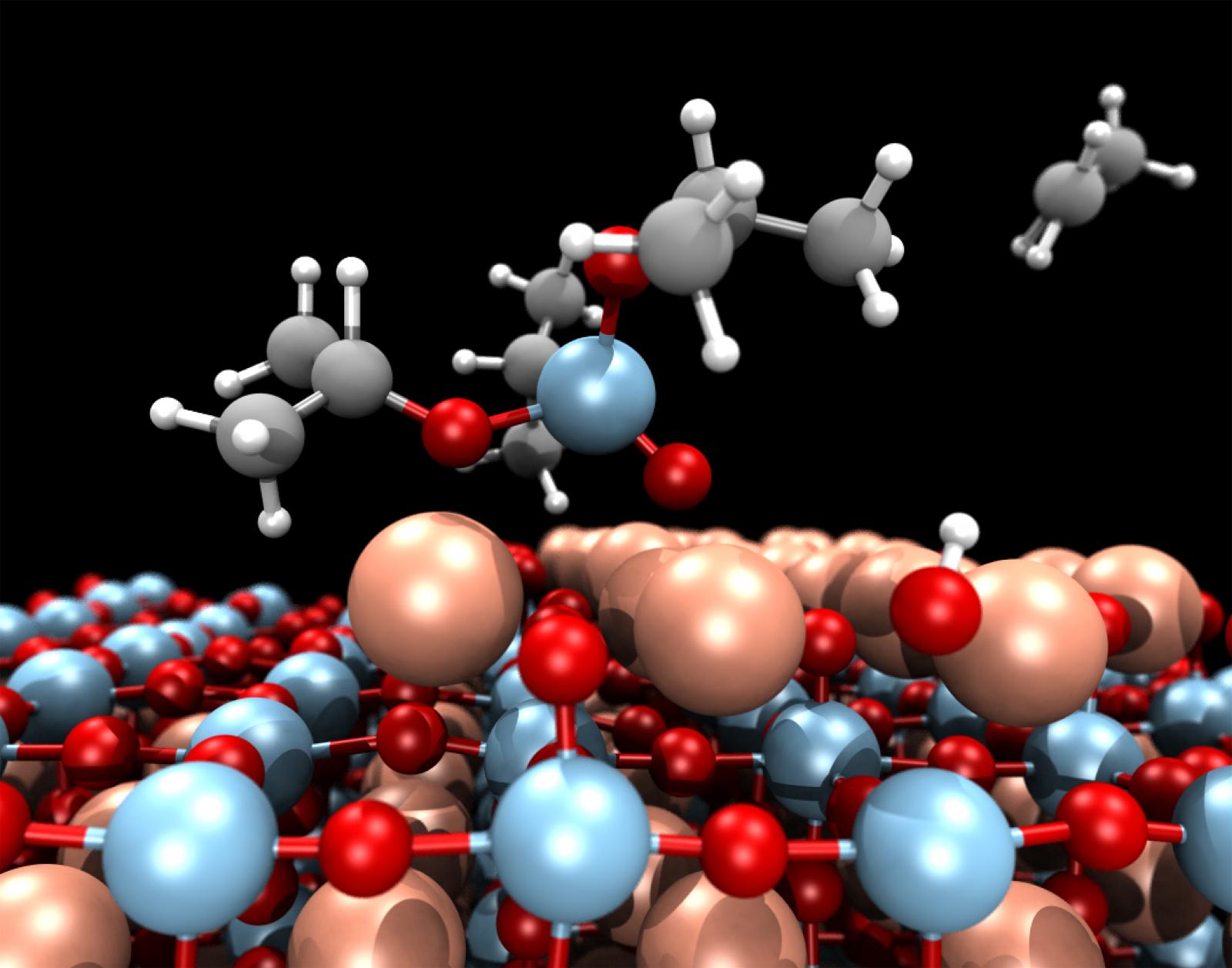
"Electrochemical charging near a pseudocapacitive electrode surface"
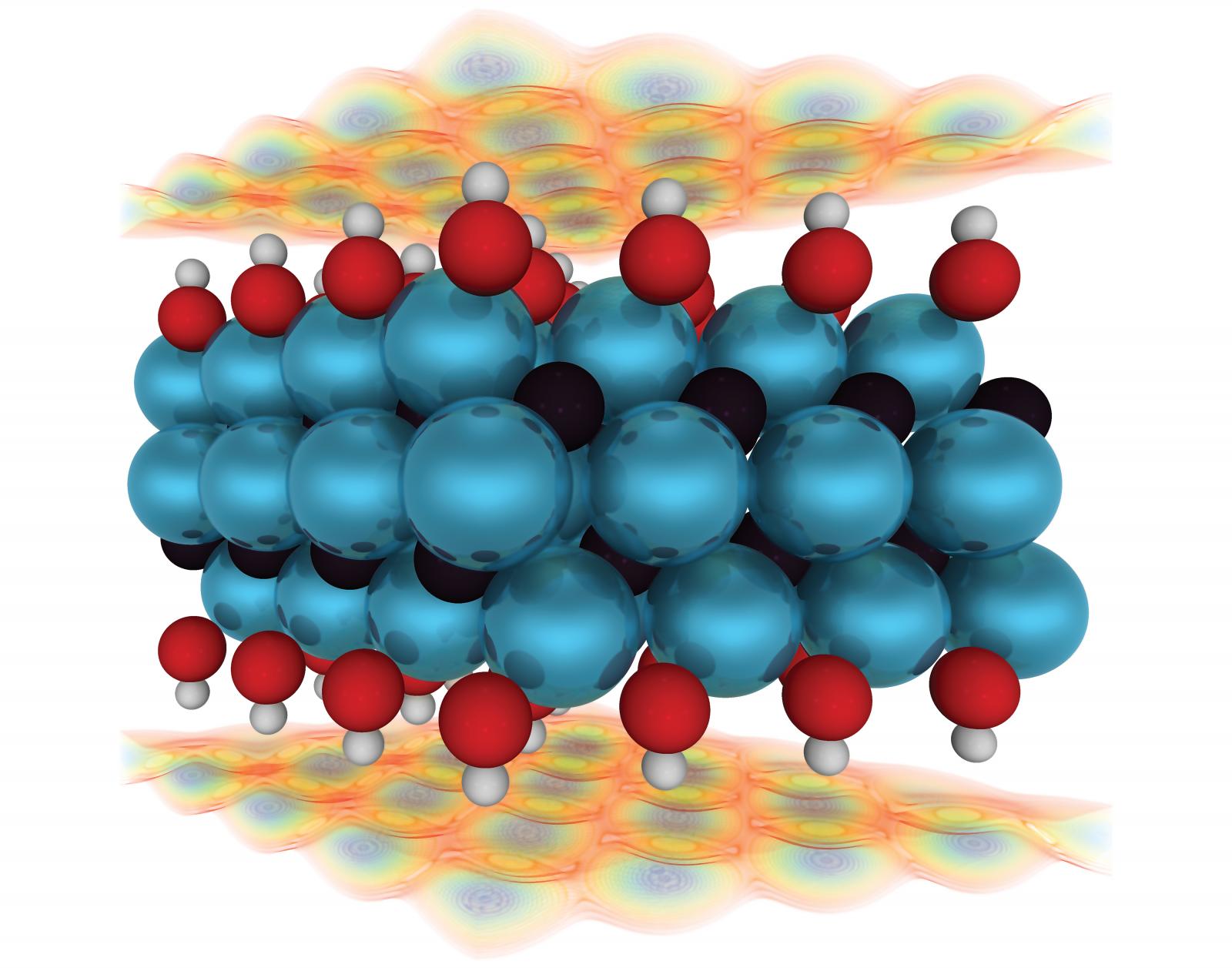
Artist's Statement: With the growing needs for cleaner energy storage and conversion, realistic models of energy storage materials are needed to aid in the design of high-performance energy storage devices. Specifically, there is a need for high-power devices for use in electric/fuel cell vehicles. First-principles calculations with continuum solvent models allow us to model these materials systems in realistic conditions. The system pictured is a Ti3C2O2 pseudocapacitive electrode, suitable for high-power transportation applications. As the electrode charges, the response of the ions in the solvent, shown as colored contours above and below the electrode surface, is related to the capacitance of the electrode.
Entry #27
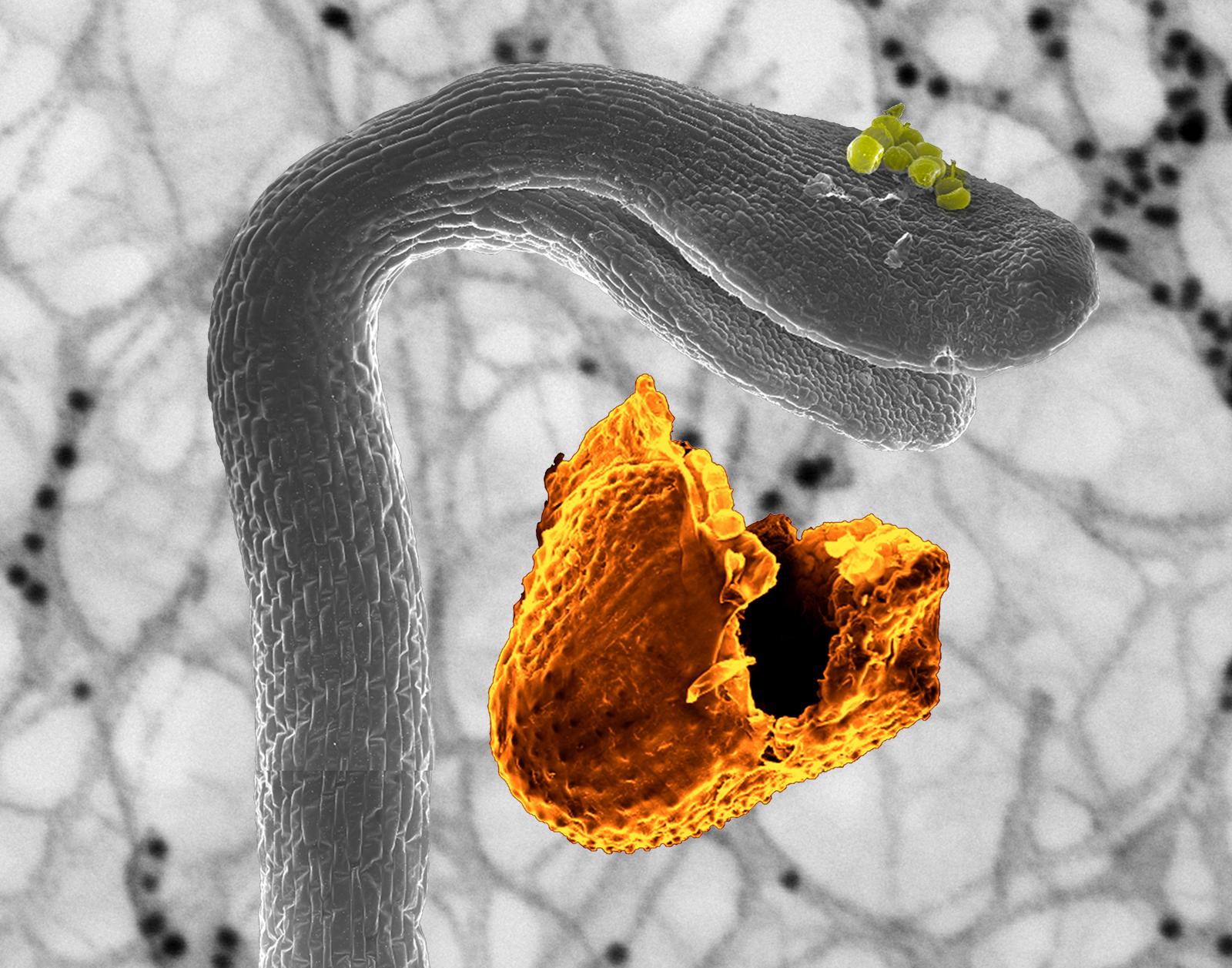
"Scanning electron micrograph of an etiolated Arabidopsis seedling"
Artist's Statement: A scanning electron micrograph of an etiolated Arabidopsis seedling (grayscale) growing in the darkness, just after it has shed its seed coat (orange). This electron micrograph is superimposed on a photograph of protein conjugated nano-gold labeled on peeled Arabidopsis seedings epidermal cell wall. This is the moment right before it would break free from the earth and reach toward the sunshine.
Entry #28
"The Silver Albacore"
Artist's Statement: A fun part of any cloudy day is always looking in the sky and trying to find figures in the clouds with friends. I compare the nanocubes I synthesize to looking at cloud formations; If you look long enough, you can see something fun in every single sample. The image was created with silver nanocubes, capped with polyvinylpyrrolidone on a silica wafer.